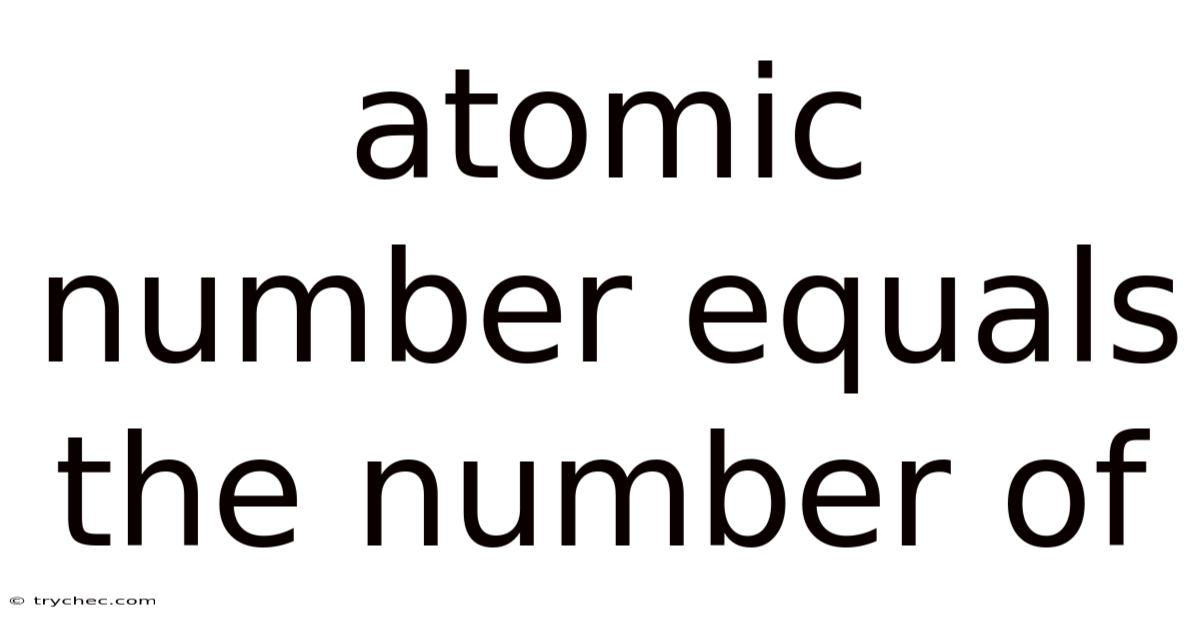The atomic number, a fundamental concept in chemistry and physics, serves as a unique identifier for a chemical element. It reveals a crucial aspect of an atom's structure and behavior, directly influencing its chemical properties. This number, assigned to each element, is equal to the number of protons found in the nucleus of an atom of that element.
The Significance of Atomic Number
The atomic number, often denoted by the symbol Z, holds critical importance for several reasons:
- Element Identification: The atomic number unambiguously identifies an element. Each element has a unique atomic number, ensuring that no two elements share the same number of protons.
- Periodic Table Organization: The periodic table, a cornerstone of chemistry, is organized based on increasing atomic numbers. This arrangement reflects the periodic trends in the elements' properties, making it easier to predict and understand their behavior.
- Chemical Properties: The number of protons in an atom dictates the number of electrons in a neutral atom. The arrangement of these electrons, particularly the valence electrons (those in the outermost shell), determines how an atom interacts with other atoms, thus governing its chemical properties.
- Nuclear Physics: The atomic number is a key parameter in nuclear physics. It helps in understanding the structure of the nucleus, nuclear reactions, and the stability of isotopes.
Historical Development
The concept of atomic number evolved over time, intertwined with the development of atomic theory and the understanding of subatomic particles:
- Early Ideas: In the 19th century, scientists recognized that elements had distinct properties, but the underlying reason remained unclear. John Dalton proposed the concept of atoms as indivisible units, but the idea of atomic number was yet to emerge.
- Mendeleev's Periodic Table: Dmitri Mendeleev arranged elements in a periodic table based on their atomic weights, noticing recurring patterns in their properties. Still, some elements were out of order based on their properties, indicating that atomic weight was not the fundamental organizing principle.
- Discovery of the Electron: J.J. Thomson's discovery of the electron in 1897 revealed that atoms were not indivisible, paving the way for understanding the internal structure of atoms.
- Rutherford's Model: Ernest Rutherford's gold foil experiment in 1911 led to the development of the nuclear model of the atom. This model proposed that an atom consists of a small, dense, positively charged nucleus surrounded by negatively charged electrons.
- Moseley's Contribution: Henry Moseley's work in 1913, using X-ray spectroscopy, demonstrated a systematic relationship between the wavelength of X-rays emitted by an element and its position in the periodic table. He concluded that the atomic number, which he equated to the number of protons in the nucleus, was the fundamental property that determined an element's identity and its position in the periodic table.
Determining the Atomic Number
The atomic number can be determined through various experimental techniques:
- X-ray Spectroscopy: Moseley's method involves bombarding a sample of an element with high-energy electrons, causing it to emit X-rays. The wavelengths of these X-rays are characteristic of the element and can be used to determine its atomic number.
- Mass Spectrometry: Mass spectrometry separates ions based on their mass-to-charge ratio. By analyzing the isotopic composition of an element, the number of protons (and hence the atomic number) can be deduced.
- Nuclear Reactions: Nuclear reactions involve changes in the nucleus of an atom. By studying these reactions and the particles emitted, the atomic number of the resulting element can be determined.
Atomic Number and Isotopes
Isotopes are atoms of the same element that have the same number of protons (same atomic number) but different numbers of neutrons. This difference in neutron number leads to variations in the atomic mass of the isotopes.
- Example: Carbon has an atomic number of 6, meaning every carbon atom has 6 protons. Still, carbon exists in nature as isotopes with different numbers of neutrons:
- Carbon-12 (*¹²C*) has 6 protons and 6 neutrons.
- Carbon-13 (*¹³C*) has 6 protons and 7 neutrons.
- Carbon-14 (*¹⁴C*) has 6 protons and 8 neutrons.
Atomic Number and Ions
Ions are atoms that have gained or lost electrons, resulting in a net electrical charge. The number of protons in an ion remains the same as in the neutral atom, so the atomic number does not change Easy to understand, harder to ignore..
- Cations: Positive ions, formed when an atom loses electrons.
- Anions: Negative ions, formed when an atom gains electrons.
Take this: sodium (Na) has an atomic number of 11. A sodium ion (*Na⁺*) is formed when a sodium atom loses one electron. The *Na⁺* ion still has 11 protons, so its atomic number remains 11.
Quantum Numbers and Atomic Structure
Quantum numbers provide a detailed description of the properties of electrons within an atom, including their energy levels, shapes of orbitals, and spatial orientation. The atomic number matters a lot in determining the electronic configuration of an atom, which in turn influences its quantum numbers.
- Principal Quantum Number (n): Describes the energy level of an electron. Higher values of n correspond to higher energy levels and greater distance from the nucleus.
- Azimuthal Quantum Number (l): Describes the shape of an electron's orbital and has values ranging from 0 to n-1. l = 0, 1, and 2 correspond to s, p, and d orbitals, respectively.
- Magnetic Quantum Number (ml): Describes the orientation of an electron's orbital in space and has values ranging from -l to +l, including 0.
- Spin Quantum Number (ms): Describes the intrinsic angular momentum of an electron, which is quantized and referred to as spin. It has values of +1/2 or -1/2.
The atomic number dictates the number of electrons in a neutral atom, which determines how these electrons are distributed among the various energy levels and orbitals. This distribution, known as the electronic configuration, follows specific rules, such as the Aufbau principle and Hund's rule, which dictate the order in which electrons fill the available orbitals.
Chemical Properties and Atomic Number
The chemical properties of an element are primarily determined by its electronic configuration, specifically the number and arrangement of valence electrons. Since the atomic number dictates the number of electrons in a neutral atom, it indirectly governs the chemical behavior of the element.
- Valence Electrons: Electrons in the outermost shell of an atom are called valence electrons. These electrons are responsible for chemical bonding and determine how an atom interacts with other atoms.
- Periodic Trends: The periodic table organizes elements based on their atomic numbers, reflecting the periodic trends in their chemical properties. Elements in the same group (vertical column) have similar valence electron configurations and, therefore, exhibit similar chemical behavior.
To give you an idea, elements in Group 1 (alkali metals) have one valence electron, making them highly reactive and prone to losing that electron to form positive ions. Elements in Group 17 (halogens) have seven valence electrons, making them highly reactive and prone to gaining one electron to form negative ions And it works..
Applications of Atomic Number
The concept of atomic number has numerous practical applications in various fields:
- Material Science: Understanding the atomic number and electronic configuration of elements is essential for designing and synthesizing new materials with specific properties.
- Nuclear Medicine: Radioactive isotopes, which are characterized by their atomic number and mass number, are used in medical imaging and cancer therapy.
- Environmental Science: The atomic number helps in identifying and quantifying pollutants in the environment.
- Archaeology: Radioactive isotopes, such as carbon-14, are used for dating archaeological artifacts.
Examples of Atomic Number in Common Elements
To illustrate the concept of atomic number, here are some examples of common elements and their atomic numbers:
- Hydrogen (H): Atomic number 1
- Carbon (C): Atomic number 6
- Oxygen (O): Atomic number 8
- Sodium (Na): Atomic number 11
- Chlorine (Cl): Atomic number 17
- Iron (Fe): Atomic number 26
- Gold (Au): Atomic number 79
- Uranium (U): Atomic number 92
Challenges and Future Directions
While the concept of atomic number is well-established, there are ongoing challenges and future directions in its application and understanding:
- Superheavy Elements: Synthesizing and characterizing superheavy elements (elements with very high atomic numbers) is a challenging task. These elements are often unstable and exist for only a fraction of a second.
- Exotic Atoms: Researchers are exploring exotic atoms, which are atoms in which one or more of the usual subatomic particles (electrons, protons, or neutrons) are replaced by other particles. These exotic atoms can provide insights into fundamental physics.
- Quantum Computing: Quantum computing holds the potential to simulate the behavior of atoms and molecules with unprecedented accuracy, leading to a deeper understanding of the role of atomic number in chemical and physical processes.
The Atomic Number and the Periodic Table
The periodic table is arguably the most recognizable icon of chemistry, and its organization is fundamentally based on the atomic number. Understanding how the atomic number dictates the arrangement and trends within the periodic table is crucial for grasping the behavior of elements.
- Organization by Atomic Number: As previously noted, the elements in the periodic table are arranged in order of increasing atomic number. This arrangement was a critical improvement over earlier attempts to organize the elements by atomic weight, which led to inconsistencies.
- Periods and Groups: The periodic table is structured into periods (horizontal rows) and groups (vertical columns).
- Periods: Elements within the same period have the same number of electron shells. As you move across a period, the atomic number increases, and the elements exhibit a gradual change in properties from metallic to nonmetallic.
- Groups: Elements within the same group have the same number of valence electrons, which results in similar chemical properties. Here's one way to look at it: the alkali metals (Group 1) all have one valence electron and are highly reactive.
- Blocks: The periodic table can also be divided into blocks based on the type of subshell that is being filled with electrons. These blocks are named after the characteristic orbitals:
- s-block: Includes Groups 1 and 2, where the valence electrons occupy s orbitals.
- p-block: Includes Groups 13-18, where the valence electrons occupy p orbitals.
- d-block: Includes Groups 3-12 (transition metals), where the valence electrons occupy d orbitals.
- f-block: Includes the lanthanides and actinides, where the valence electrons occupy f orbitals.
The arrangement of elements by atomic number in the periodic table allows for easy identification of elements with similar properties and the prediction of their behavior in chemical reactions.
The Role of Atomic Number in Chemical Bonding
Chemical bonding, the process by which atoms combine to form molecules and compounds, is fundamentally governed by the interactions of valence electrons. Since the atomic number determines the number of electrons in an atom, it indirectly is key here in chemical bonding.
- Ionic Bonding: In ionic bonding, electrons are transferred from one atom to another, resulting in the formation of ions (charged particles). The atomic number determines the number of electrons an atom needs to gain or lose to achieve a stable electron configuration (usually a full outer shell). Take this: sodium (Na, atomic number 11) readily loses one electron to form a *Na⁺* ion, while chlorine (Cl, atomic number 17) readily gains one electron to form a *Cl⁻* ion. These ions then attract each other due to their opposite charges, forming an ionic bond in sodium chloride (NaCl).
- Covalent Bonding: In covalent bonding, atoms share electrons to achieve a stable electron configuration. The number of electrons an atom needs to share is determined by its atomic number and the number of valence electrons. As an example, carbon (C, atomic number 6) has four valence electrons and can form four covalent bonds with other atoms, as seen in methane (CH₄).
- Metallic Bonding: In metallic bonding, electrons are delocalized among a lattice of metal atoms. The atomic number determines the number of valence electrons that are available for delocalization, which influences the strength and properties of the metallic bond.
Advanced Concepts Related to Atomic Number
Beyond the basic definition and applications, the atomic number is also relevant in more advanced concepts in chemistry and physics:
- Effective Nuclear Charge: The effective nuclear charge (*Zeff*) is the net positive charge experienced by an electron in a multi-electron atom. It is less than the actual nuclear charge (atomic number) due to the shielding effect of other electrons. The effective nuclear charge influences the energy levels of electrons and their interactions with the nucleus.
- Slater's Rules: Slater's rules provide a method for estimating the effective nuclear charge experienced by an electron in an atom. These rules take into account the shielding effect of other electrons in the atom.
- Aufbau Principle: The Aufbau principle states that electrons fill atomic orbitals in order of increasing energy. The order of filling is determined by the (n + l) rule, where n is the principal quantum number and l is the azimuthal quantum number. This principle helps predict the electronic configuration of an atom based on its atomic number.
- Hund's Rule: Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. This rule is based on the principle that electrons minimize their energy by maximizing their spin multiplicity.
Conclusion
The atomic number is a cornerstone concept in chemistry and physics. It serves as a unique identifier for each element, dictating the number of protons in the nucleus of an atom. This fundamental property influences the arrangement of electrons, the chemical behavior of elements, and their organization in the periodic table. From its historical development to its numerous applications, the atomic number continues to be a vital tool for understanding the structure and properties of matter That alone is useful..